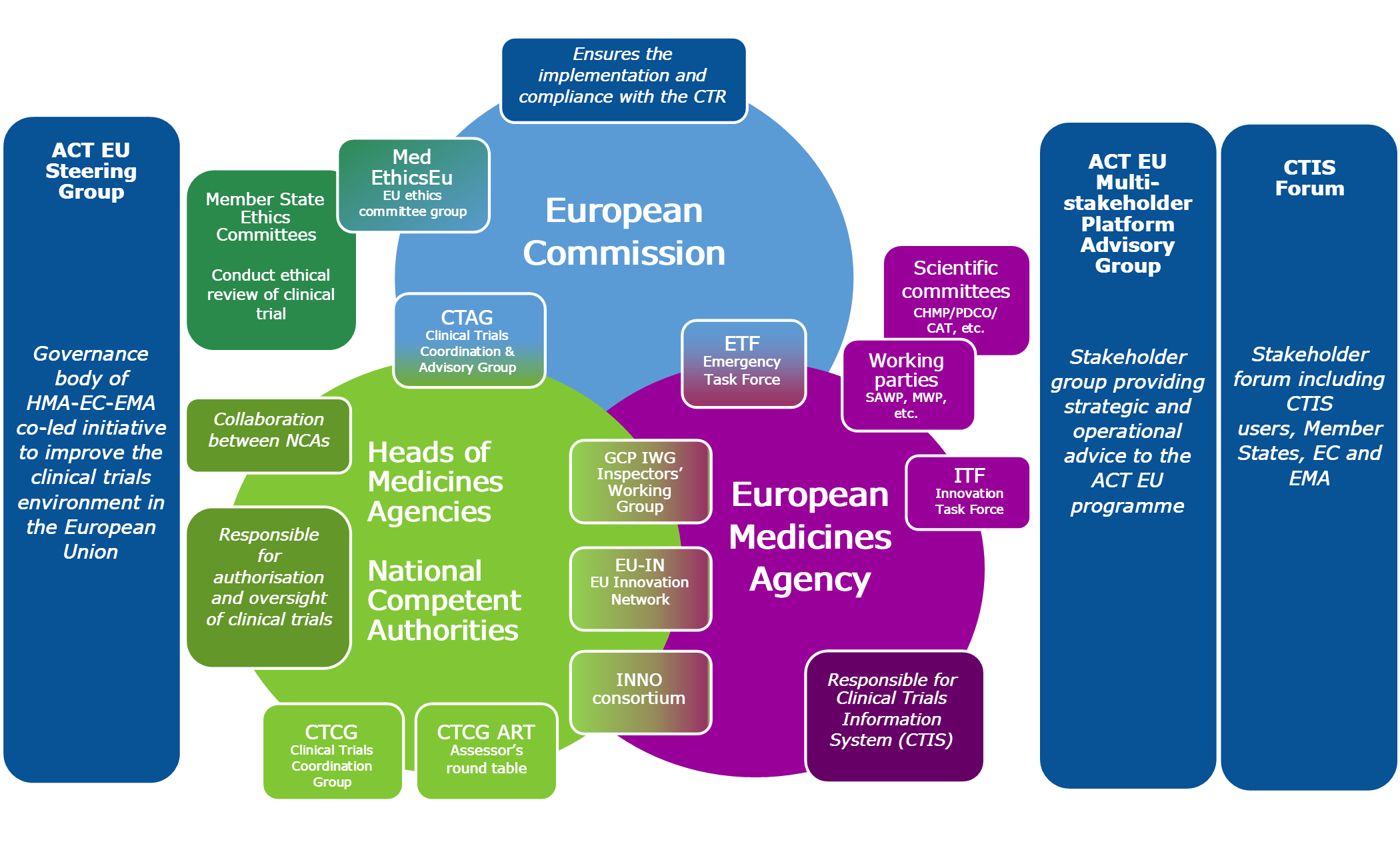
The ACT EU programme has mapped existing clinical trial activities to develop a governance rationalisation strategy.
This aims to clarify the roles and responsibilities of the various expert groups working within the European medicines regulatory network (EMRN).
The Clinical Trials Coordination Group (CTCG) is leading the Clinical Trials Regulation (CTR) Collaborate project which aims to optimise the collaboration of ethics committees and national competent authorities regarding clinical trials authorisation under the CTR. Anchoring the initiative to ACT EU enables the prioritisation of CTR related activities under the programme.

Clinical Trials Coordination and Advisory Group (CTAG)
| Anchoring | European Commission, Lead DG: SANTE – DG Health and Food Safety* Article 85 of the Clinical Trials Regulation (EU) 536/2014 (“CTR”) |
|---|---|
| Participants | CTR National Contact Points (one representative per Member State), CTCG observer, EMA observer |
| Meeting frequency | At least four times a year |
| Purpose/Mission | To contribute to the implementation of the CTR, including its implementing acts. |
| Tasks |
|
| Relevant links |
Clinical Trials Coordination Group (CTCG)
| Anchoring | Heads of Medicines Agencies working group |
|---|---|
| Participants | Working group of experts in the classification, assessment and oversight of clinical trials from National Agencies |
| Meeting frequency | Monthly and ad-hoc when necessary |
| Purpose/Mission | To contribute to increasing the attractiveness of the EU/EEA for clinical trials by promoting harmonisation and optimisation of the regulatory environment while assuring the protection of rights, safety and wellbeing of participants and the generation of robust data. |
| Tasks | |
| Relevant links | Heads of Medicines Agencies: Clinical Trials Coordination Group (hma.eu) |
CTCG assessors round table (ART)
| Anchoring | Subgroup under CTCG |
|---|---|
| Participants | Members from NCAs and ethics committees |
| Meeting frequency | Weekly, with individual topics covered once per month (pre-clinical, clinical, quality and part II related topics) with separate meetings for scientific advice. Once a month, members from ethics committees are invited to join, if not already participating. |
| Purpose/Mission | To discuss regulatory and scientific issues arising during the assessment of clinical trials, to discuss solutions, drive consistency of approaches across Member States and to provide training. |
| Tasks | Discuss solutions to questions raised by sponsors and MSs to drive harmonised approaches across NCAs and ethics committees. |
| Relevant links | Not applicable |
MedEthicsEU
| Anchoring | Special group under the European Commission, Support DG: SANTE – DG Health and Food Safety*, led by MedEthicsEU board. |
|---|---|
| Participants | Austria, Belgium, Bulgaria, Croatia, Cyprus, Czech Republic, Denmark, Estonia, Finland, France, Germany, Greece, Hungary, Iceland, Ireland, Italy, Latvia, Lithuania, Liechtenstein, Luxembourg, Malta, Netherlands, Norway, Poland, Portugal, Romania, Slovenia, Slovakia, Spain, Sweden |
| Meeting frequency | Monthly and ad-hoc when necessary |
| Purpose/Mission | To strengthen collaboration between the medical research ethics committees reviewing clinical trials applications falling under the Clinical Trials Regulation (CTR), performance study applications falling under the In Vitro Diagnostics Regulation (IVDR) and clinical investigation applications falling under Medical Device Regulation (MDR) in the EU/EEA Member States. |
| Tasks |
|
| Relevant links | MedEthicsEU - European Commission (europa.eu) |
INNO Steering Group
| Anchoring | Heads of Medicines Agencies (HMA) voluntary discussion forum |
|---|---|
| Participants |
The Steering group consists of co-chairs of the groups involved to enhance the communication between the group. The chair is provided by the HMA. |
| Meeting frequency | Ad hoc in addition to an annual meeting |
| Purpose/Mission | To enhance the dialogue and communication between the groups giving scientific and/or regulatory advice to innovators; to set the strategy and vision for collaboration and plan the group’s annual meetings. |
| Tasks | |
| Relevant links | Not applicable |
GCP Inspectors’ Working Group (GCP IWG)
| Anchoring | Heads of Medicines Agencies Article 57(1)(i) of Regulation (EC) No 726/2004 |
|---|---|
| Participants | Representatives of the good clinical practice (GCP) inspectorates of the European Economic Area Member States, and the EMA Inspections Office. Observers from the European Commission, the World Health Organisation, Swissmedic and EU enlargement countries. The chair is provided by the EMA Inspections office. |
| Meeting frequency | Four times per year |
| Purpose/Mission | To promote harmonisation and co-ordination of GCP related activities at community level to discuss and align positions in relation to compliance issues. |
| Tasks |
|
| Relevant links | Good Clinical Practice Inspectors Working Group | European Medicines Agency (europa.eu) |
ACT EU Steering Group (ACT EU SG)
| Anchoring | Initiative co-led by the European Commission, Heads of Medicines Agencies (HMA) and the European Medicines Agency (EMA). The Steering Group reports to the EMA Management Board and HMA. |
|---|---|
| Participants | Members from the European Commission, EMA, HMA, and the Network Portfolio Advisory Group (NPAG). The chair is provided by the European Commission. |
| Meeting frequency | Monthly |
| Purpose/Mission | To accelerate clinical trials in the EU/EEA by creating a favourable environment for clinical research. |
| Tasks |
|
| Relevant links | ACT EU website: Governance |
EU Innovation Network (EU-IN)
| Anchoring | Working group of European Medicines Agency (EMA) and the Heads of Medicines Agencies (HMA) |
|---|---|
| Participants | Composed by representatives of NCA innovation offices as well the EMA Innovation Task Force (ITF). |
| Meeting frequency | 11 virtual meetings per year. 1 face to face meetings per year in the context of a meeting on innovation with relevant stakeholders including SMEs and academic groups. |
| Purpose/Mission | To facilitate the development of innovative medicines and technologies for drug development including innovative unprecedented concepts of clinical trial design and methodologies by addressing gaps in early regulatory support to innovation. |
| Tasks |
|
| Relevant links | EU Innovation Network (EU-IN) | European Medicines Agency (europa.eu) |
Scientific advice working party (SAWP)
| Anchoring | Standing working party established by the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA). |
|---|---|
| Participants | Multidisciplinary group composed of European experts, including three members of the Committee for Orphan Medicinal Products (COMP), three members of the Paediatric Committee (PDCO), three members of the Committee for Advanced Therapies (CAT) and one member of the Pharmacovigilance Risk Assessment Committee (PRAC). |
| Meeting frequency | 11 meetings per year |
| Purpose/Mission | To optimise research and development, reduce uncertainties in regulatory outcomes, and accelerate time to approval of a marketing authorisation application. |
| Tasks | SAWP coordinates the provision of scientific advice and protocol assistance. It brings an integrated view forward to the CHMP and the COMP on:
|
| Relevant links | Scientific Advice Working Party | European Medicines Agency (europa.eu) |
Methodology working party (MWP)
| Anchoring | Working party of the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) |
|---|---|
| Participants | The MWP is composed of European experts nominated by CHMP members. |
| Meeting frequency | Monthly |
| Purpose/Mission | To leverage cross-disciplinary expertise to support methodological innovation in global drug development, and support advice and interpretation of complex methodology across (clinical) drug development. |
| Tasks |
|
| Relevant links | Methodology Working Party | European Medicines Agency (europa.eu) |
Innovation Task Force (ITF)
| Anchoring | EMA multidisciplinary horizontal cross-sectorial group |
|---|---|
| Participants | ITF members are scientific and legal administrators appointed from all areas of the EMA. The ITF network includes all EU Network and additional experts as relevant for the topic discussion at hand (enrichment function of EMA expert pool). |
| Meeting frequency | |
| Purpose/Mission | To provide early scientific, legal and regulatory support to global innovative medicine developments intended for the EU market/ regulatory system with a particular focus on emerging therapies and technologies. Preparatory early entry door for innovation in medicines. |
| Tasks |
|
| Relevant links | Supporting innovation | European Medicines Agency (europa.eu) |
Emergency Task Force (ETF)
| Anchoring | Established by EMA in accordance with the Regulation on EMA's Reinforced Role (Regulation (EU) 2022/123). The ETF took over the activities of the COVID-19 EMA Pandemic Task Force that EMA convened in March 2020 to address the COVID-19 pandemic. |
|---|---|
| Participants |
|
| Meeting frequency | Adapted to the level of activity required with ad hoc meetings as needed. |
| Purpose/Mission | To ensure that high quality, safe and efficacious medicinal products, which have the potential to address public health emergencies, can be developed and made available within the European Union as soon as possible during public health emergencies. |
| Tasks |
|
| Relevant links | Emergency Task Force (ETF) | European Medicines Agency (europa.eu) |
Clinical Trials Information System Forum (CTIS Forum)
| Anchoring | The CTIS Forum was formed after the go live of the Clinical Trials Regulation (EU) No. 536/2014 in 2022. |
|---|---|
| Participants | The CTIS Forum includes CTIS users and experts from Member States, EU sponsors (academic, industry), from Clinical Research Organisations (CROs), from patients (Patients/Consumers Working Party (PCWP) representatives) and from healthcare professionals (Healthcare Professionals Working Party (HCPWP) representatives). |
| Meeting frequency | At least every 3 months |
| Purpose/Mission | To allow direct exchanges of information, to discuss user experiences and to inform about CTIS operations and delivery, training and communication as well as other relevant topics in the context of CTIS. |
| Tasks | It aims to promote dialogue; facilitate the flow of information between Member States, external stakeholders, the Agency and the European Commission; and to escalate general business operational aspects of CTIS to CTIS MS PO Experts Group and, if needed, to the ACT EU Steering Group. |
| Relevant links | Not applicable |
ACT EU Multi-stakeholder Platform Advisory Group (MSP AG)
| Anchoring | Established as part of the ACT EU initiative co-led by European Commission, Heads of Medicines Agencies and European Medicines Agency. The MSP AG reports to the ACT EU Steering Group. |
|---|---|
| Participants | Representatives of:
Two ethics committee representatives and ACT EU regulatory partners, actively participate in the meetings. |
| Meeting frequency | Quarterly meetings, with the possibility of ad hoc meetings as needed |
| Purpose/Mission | The MSP AG brings together key stakeholder groups who are directly impacted by clinical trial-related activities in the EU to increase collaboration, build mutual trust and with the ultimate goal of improving the EU clinical trials landscape. The MSP AG provides insight into the perspectives and needs of the different stakeholder groups and allows stakeholders to provide strategic and operational advice regarding ACT EU. |
| Tasks |
|
| Relevant links | ACT EU website: Multi-stakeholder platform |
News

The ACT EU workplan for 2026-2027 outlines the initiative's revised objectives and areas of focus, informed by feedback from stakeholders.

COMBINE “all-in-one” coordinated assessment pilot: call open for expressions of interest until 31 August 2025.

The ACT EU workplan for 2025-2026 outlines the initiative's areas of focus, which reflect feedback from stakeholders.